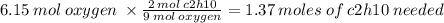Given the following balanced chemical equation:
2C2H10 + 902 +4CO2 + 10H2O
How many moles of...

Chemistry, 14.04.2021 14:00 genyjoannerubiera
Given the following balanced chemical equation:
2C2H10 + 902 +4CO2 + 10H2O
How many moles of C2H10 are needed to completely react with 6.15
moles of oxygen?
O 1.37 mol
2 mol
0277 mol

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:00
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

Chemistry, 22.06.2019 13:30
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2
You know the right answer?
Questions





English, 03.12.2019 21:31


Mathematics, 03.12.2019 21:31



Mathematics, 03.12.2019 21:31


Chemistry, 03.12.2019 21:31

Biology, 03.12.2019 21:31


English, 03.12.2019 21:31


Mathematics, 03.12.2019 21:31

Social Studies, 03.12.2019 21:31