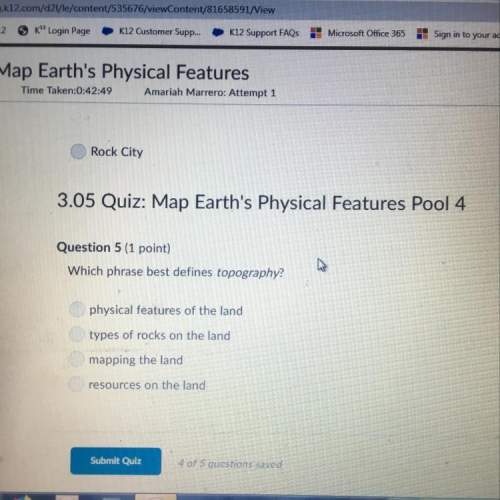
Identify the correct statement regarding the strength of chemical bonds.
Strong bonds occur with high temperature and weak bonds with low temperature.
If the bond energy between atoms is very weak then the bond between the atoms is strong.
Strong bonds form with large atoms and weak bonds with small atoms.
If the bond energy between atoms is very high then the bond between the atoms is strong.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1

Chemistry, 22.06.2019 21:30
An atomic nucleus is composed ofa)protons.b)protons and neutrons.c)protons and electrons.d)protons, neutrons, and electrons.
Answers: 1

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
Identify the correct statement regarding the strength of chemical bonds.
Strong bonds occur with hi...
Questions

Mathematics, 20.10.2020 05:01

Social Studies, 20.10.2020 05:01

History, 20.10.2020 05:01

Mathematics, 20.10.2020 05:01


Mathematics, 20.10.2020 05:01

Geography, 20.10.2020 05:01

Social Studies, 20.10.2020 05:01




Mathematics, 20.10.2020 05:01

Biology, 20.10.2020 05:01





History, 20.10.2020 05:01

Mathematics, 20.10.2020 05:01

Physics, 20.10.2020 05:01