
Chemistry, 14.04.2021 18:10 2022mcwhirterbrendan
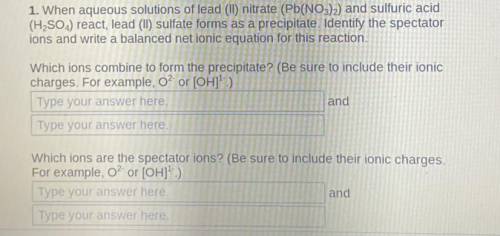
1. When aqueous solutions of lead (II) nitrate (Pb(NO3)2) and sulfuric acid
(H2SO4) react, lead (II) sulfate forms as a precipitate.
Identify the spectator
ions and write a balanced net ionic equation for this reaction.
Which ions combine to form the precipitate? (Be sure to include their ionic
charges. For example, o? or [OH]1- )
Which ions are the spectator ions?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1
You know the right answer?
1. When aqueous solutions of lead (II) nitrate (Pb(NO3)2) and sulfuric acid
(H2SO4) react, lead (II...
Questions

English, 25.02.2020 01:05

Mathematics, 25.02.2020 01:05




Mathematics, 25.02.2020 01:05

Computers and Technology, 25.02.2020 01:05

Mathematics, 25.02.2020 01:05

History, 25.02.2020 01:05




Mathematics, 25.02.2020 01:05

English, 25.02.2020 01:05



English, 25.02.2020 01:05

Mathematics, 25.02.2020 01:05




