Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 23.06.2019 00:00
How many atoms or molecules are there in a mole of a substance?
Answers: 1

Chemistry, 23.06.2019 09:00
The concentration of ionic substances is important for the heart to beat. your heart responds to electrical impulses that travel through heart cells that are made up mostly of water. which properties of ionic compounds are important to support this function? solubility in water conductivity crystalline melting point
Answers: 3

Chemistry, 23.06.2019 09:30
What is the best describtion of the side of the moon that faces earth?
Answers: 1
You know the right answer?
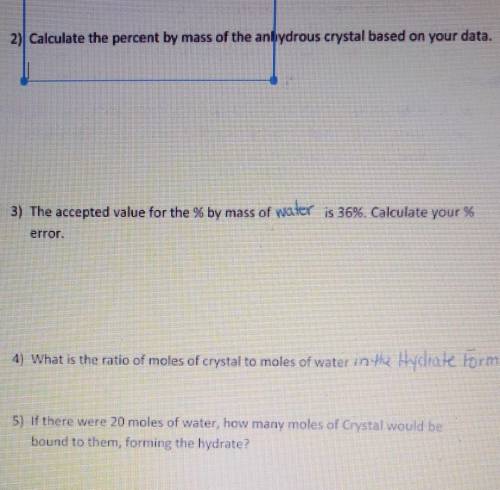
BASED OFF LAST QUESTION
...
...
Questions

Mathematics, 22.02.2021 23:30




Advanced Placement (AP), 22.02.2021 23:30


Physics, 22.02.2021 23:30

Mathematics, 22.02.2021 23:30

English, 22.02.2021 23:30

English, 22.02.2021 23:30



Mathematics, 22.02.2021 23:30


Health, 22.02.2021 23:30

Mathematics, 22.02.2021 23:30

English, 22.02.2021 23:30

Mathematics, 22.02.2021 23:30


English, 22.02.2021 23:30