
Chemistry, 16.04.2021 01:00 davienwatson8
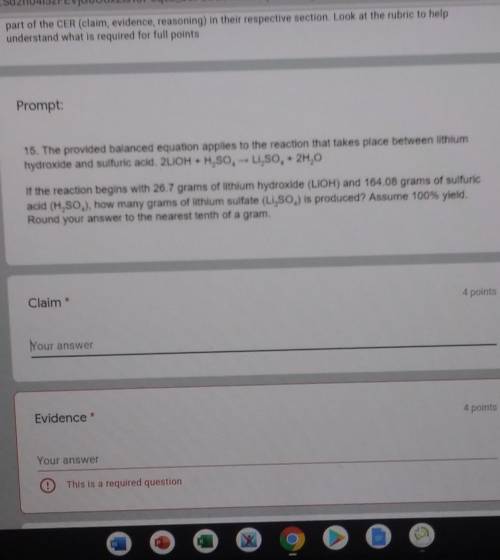
The provided balanced equation applies to the reaction that takes place between lithium, hydroxide and sulfuric acid. 2LiOH + H2SO4 --> Li2SO4 + 2H2O. If the reaction begins with 26.7 grams of lithium hydroxide (LiOH) and 164.048 grams of sulfuric acid (H2SO4), how many grams of lithium sulfate (Li2SO4) is produced? Assume 100% yield. Round your answer to the nearest tenth of a gram

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2

Chemistry, 23.06.2019 03:30
Astudent uses universal ph paper to find the ph of three solutions . solution a has a ph of 5 solution b has a ph of 11 and solution c has a ph of 7 identify which solution is acidic which solution is neutral and which solution is basic
Answers: 1

Chemistry, 23.06.2019 07:30
Using this reversible reaction, answer the questions below: n2o4 2no2 (colorless) (reddish-brown) -as the temperature increased, what happened to the n2o4 concentration? -was the formation of reactants or products favored by the addition of heat? -which reaction is exothermic? right to left or left to right? -if the change of enthalpy of this reaction when proceeding left to right is 14 kcal, which chemical equation is correct? n2o4 2no2 + 14 kcal n2o4 2no2, hr = +14 kcal n2o4 + 14 kcal 2no2 n2o4 2no2, hr = -14 kcal
Answers: 1
You know the right answer?
The provided balanced equation applies to the reaction that takes place between lithium, hydroxide a...
Questions



Mathematics, 12.01.2021 03:30

English, 12.01.2021 03:30



Mathematics, 12.01.2021 03:30







Engineering, 12.01.2021 03:30

English, 12.01.2021 03:30


History, 12.01.2021 03:30


Health, 12.01.2021 03:30

Mathematics, 12.01.2021 03:30



