
Chemistry, 17.04.2021 01:00 Albraaalouda
is 0.6745 g sample of an unknown acid HX reacts with 41.75 mL of KOH solution for complete neutralization. What is the molarity of the KOH solution
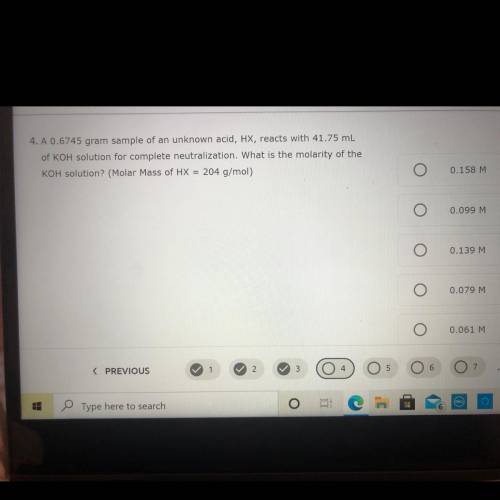

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 05:30
The climate of the continental united states is generally 1. tropical 2. temperate 3. arctic 4. highland
Answers: 1

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 23.06.2019 04:10
In an experiment, 45g of silicon tetrachloride are treated with 45ml of water. what is the theoretical yield in grams of hcl
Answers: 3
You know the right answer?
is 0.6745 g sample of an unknown acid HX reacts with 41.75 mL of KOH solution for complete neutraliz...
Questions




Mathematics, 16.12.2019 19:31


Mathematics, 16.12.2019 19:31

History, 16.12.2019 19:31

Biology, 16.12.2019 19:31



English, 16.12.2019 19:31


History, 16.12.2019 19:31

English, 16.12.2019 19:31


Mathematics, 16.12.2019 19:31


Mathematics, 16.12.2019 19:31

Biology, 16.12.2019 19:31

Mathematics, 16.12.2019 19:31



