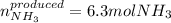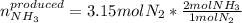Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3


Chemistry, 22.06.2019 07:30
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
You know the right answer?
Use the following chemical equation, how many moles of ammonia (NH 3 ) produced when 3.15 moles of n...
Questions





Social Studies, 22.05.2020 19:06

Mathematics, 22.05.2020 19:06



Mathematics, 22.05.2020 19:06

Social Studies, 22.05.2020 19:06

Physics, 22.05.2020 19:06

English, 22.05.2020 19:06

Mathematics, 22.05.2020 19:06


Mathematics, 22.05.2020 19:06


Mathematics, 22.05.2020 19:06



Arts, 22.05.2020 19:06