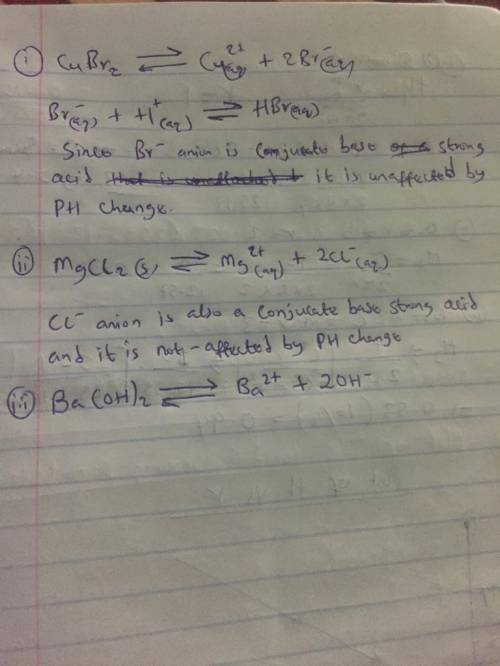Chemistry, 19.04.2021 16:10 Albertrami2251
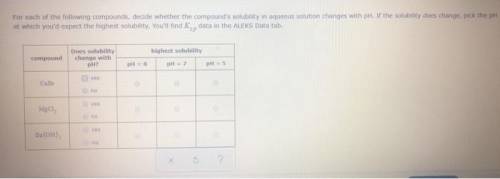
CaCO3 For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with pH. If the solubility does change, pick the pH at which you'd expect the highest solubility. You'll find data in the ALEKS Data tab. compound Does solubility change with pH? highest solubility pH = 5 pH = 6 pH = 7 yes no yes no yes no g

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Drug abuse will not lead to physical and psychological dependence. true or false ?
Answers: 2

Chemistry, 23.06.2019 01:00
Chromium(iii) sulfate is a transition metal compound containing the metal chromium and the polyatomic ion sulfate. the oxidation state of chromium in this compound is , and the chemical formula of the compound is ( ) . reset next
Answers: 3

Chemistry, 23.06.2019 02:00
To calculate the molarity of a solution, you need to know the moles of solute and the
Answers: 2

You know the right answer?
CaCO3 For each of the following compounds, decide whether the compound's solubility in aqueous solut...
Questions





History, 20.09.2019 07:10

Chemistry, 20.09.2019 07:10

Mathematics, 20.09.2019 07:10



English, 20.09.2019 07:10


Mathematics, 20.09.2019 07:10

Chemistry, 20.09.2019 07:10




Mathematics, 20.09.2019 07:10

World Languages, 20.09.2019 07:10


Mathematics, 20.09.2019 07:10