
Chemistry, 22.11.2019 22:31 lilquongohard
Write the complete balanced equation for the following reaction:
c4h10 + o2 yields co2 + h2o

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3

Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2
You know the right answer?
Write the complete balanced equation for the following reaction:
c4h10 + o2 yields co2...
c4h10 + o2 yields co2...
Questions



English, 07.10.2019 08:01


Mathematics, 07.10.2019 08:01

Mathematics, 07.10.2019 08:01



Mathematics, 07.10.2019 08:01

Chemistry, 07.10.2019 08:01

Arts, 07.10.2019 08:01

Advanced Placement (AP), 07.10.2019 08:01


Mathematics, 07.10.2019 08:01


Health, 07.10.2019 08:01

Mathematics, 07.10.2019 08:01

Arts, 07.10.2019 08:01

Mathematics, 07.10.2019 08:01

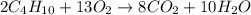
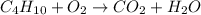
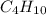 , coefficient '13' is put before the
, coefficient '13' is put before the 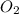 , coefficient '8' is put before the
, coefficient '8' is put before the 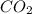 and coefficient '10' is put before the
and coefficient '10' is put before the 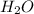 and we get the balanced chemical equation.
and we get the balanced chemical equation.

