Option (B) is the correct answer.
Explanation:
Entropy is defined as the degree of disorder or randomness. This means that when particles of a matter move rapidly and continuously from one place to another then this shows entropy of the matter is high.
For example, particles of a gas are weakly held and hence, they rapidly collide and show high entropy.
Whereas in solids, particles are held together by strong intermolecular forces of attraction. Thus, they are not moving and hence in solids entropy is the lowest.
Thus, we can conclude that out of the given options, 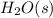 has the lowest entropy.
has the lowest entropy.



























