Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 17:00
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 22.06.2019 23:00
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2
You know the right answer?
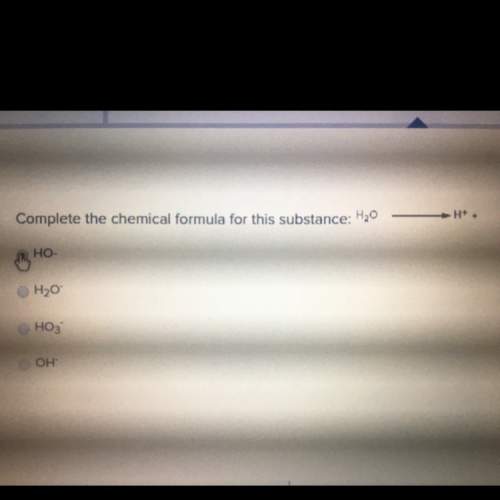
Can anyone me with this? the picture may or may not show up on your screen and if not i can probab...
Questions

History, 17.05.2021 19:30




Physics, 17.05.2021 19:30

Mathematics, 17.05.2021 19:30

Business, 17.05.2021 19:30


Mathematics, 17.05.2021 19:30

English, 17.05.2021 19:30

Social Studies, 17.05.2021 19:30




Arts, 17.05.2021 19:30



Social Studies, 17.05.2021 19:30

Biology, 17.05.2021 19:30