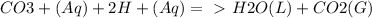Chemistry, 21.09.2019 08:50 yarrito20011307
Every antacid contains one or more ingredients capable of reacting with excess stomach acid (hcl). the essential neutralization products are co2 and/or h2o. write net ionic equations to represent the neutralizing action of the following popular antacids:
-rolaids
-maalox
-tums
-milk of magnesia

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 23.06.2019 02:00
Why does ammonia, nh3, behave as a base when it reacts with an acid? z
Answers: 2

Chemistry, 23.06.2019 16:00
Why is it important for scientists to replicate each other’s experiments? to determine if important scientific results are repeatable to the research of other scientists to determine if slight alterations in the experiment can affect the result to further their own research
Answers: 2
You know the right answer?
Every antacid contains one or more ingredients capable of reacting with excess stomach acid (hcl). t...
Questions



History, 01.08.2019 02:00


Geography, 01.08.2019 02:00


History, 01.08.2019 02:00




World Languages, 01.08.2019 02:00

History, 01.08.2019 02:00


Business, 01.08.2019 02:00






Biology, 01.08.2019 02:00