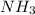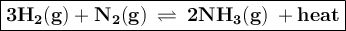Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2
You know the right answer?
Diatomic hydrogen gas and diatomic nitrogen gas react spontaneously to form a gaseous product. give...
Questions

Biology, 08.04.2020 18:37


Mathematics, 08.04.2020 18:37





History, 08.04.2020 18:37


Health, 08.04.2020 18:37




Mathematics, 08.04.2020 18:37

Physics, 08.04.2020 18:37

English, 08.04.2020 18:37


History, 08.04.2020 18:37