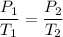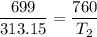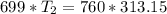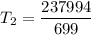Chemistry, 29.09.2019 15:30 kitttimothy55
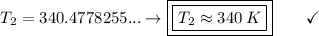
An unknown gas has a pressure of 699.0 mm hg at 40.0 c. what is the temperature at standard pressure?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
You know the right answer?
An unknown gas has a pressure of 699.0 mm hg at 40.0 c. what is the temperature at standard pressure...
Questions


Mathematics, 24.01.2020 07:31

Mathematics, 24.01.2020 07:31


History, 24.01.2020 07:31

Mathematics, 24.01.2020 07:31

Mathematics, 24.01.2020 07:31

Arts, 24.01.2020 07:31


Mathematics, 24.01.2020 07:31


English, 24.01.2020 07:31

Mathematics, 24.01.2020 07:31

Mathematics, 24.01.2020 07:31



Mathematics, 24.01.2020 07:31