Chemistry, 04.02.2020 03:03 bryantpropst1395
How many grams of zinc metal will react completely with 5.4 liters of 2.8 m hcl? show all of the work needed to solve this problem. zn (s) + 2 hcl (aq) yields zncl2 (aq) + h2 (g)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 23.06.2019 20:00
What is the empirical formula if you have 81.82% carbon and 18.18% hydrogen?
Answers: 1
You know the right answer?
How many grams of zinc metal will react completely with 5.4 liters of 2.8 m hcl? show all of the wo...
Questions



Mathematics, 30.10.2020 20:50

Arts, 30.10.2020 20:50

Mathematics, 30.10.2020 20:50


Mathematics, 30.10.2020 20:50

Mathematics, 30.10.2020 20:50

Mathematics, 30.10.2020 20:50

Business, 30.10.2020 20:50



Mathematics, 30.10.2020 20:50



Mathematics, 30.10.2020 20:50

Mathematics, 30.10.2020 20:50


Mathematics, 30.10.2020 20:50

Chemistry, 30.10.2020 20:50

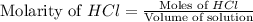
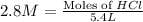
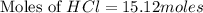
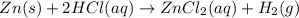
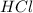 react to with 1 mole of Zn
react to with 1 mole of Zn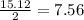 moles of Zn
moles of Zn