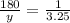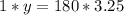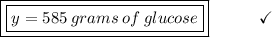Chemistry, 01.02.2020 03:44 kameronstebbins
Determine the mass in grams of 3.25 mols of glucose(c6h12o6)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1

Chemistry, 23.06.2019 09:30
Where are the noble gases located in the periodic table? a. in the center b. on the left side c. in the upper right corner d. on the far right side
Answers: 1

Chemistry, 23.06.2019 10:30
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle. if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3.2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1
You know the right answer?
Determine the mass in grams of 3.25 mols of glucose(c6h12o6)...
Questions


Chemistry, 28.01.2021 17:30

Mathematics, 28.01.2021 17:30


English, 28.01.2021 17:30

Business, 28.01.2021 17:30

Biology, 28.01.2021 17:30

Chemistry, 28.01.2021 17:30



Engineering, 28.01.2021 17:30


Mathematics, 28.01.2021 17:30