
How does the number of molecules in 1 mol of oxygen compare with the number of molecules in 1 mol of nitrogen?
a.) 1 mol of oxygen has fewer molecules.
b.) 1 mol of oxygen has more molecules.
c.) each sample has the same number of molecules.
d.) the molecules cannot be compared without knowing the mass.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1
You know the right answer?
How does the number of molecules in 1 mol of oxygen compare with the number of molecules in 1 mol of...
Questions


Computers and Technology, 16.10.2019 04:00


Mathematics, 16.10.2019 04:00


Mathematics, 16.10.2019 04:00

Mathematics, 16.10.2019 04:00


Mathematics, 16.10.2019 04:00

Mathematics, 16.10.2019 04:00

Mathematics, 16.10.2019 04:00

Biology, 16.10.2019 04:00


Mathematics, 16.10.2019 04:00

Mathematics, 16.10.2019 04:00

History, 16.10.2019 04:00

Mathematics, 16.10.2019 04:00

Social Studies, 16.10.2019 04:00

History, 16.10.2019 04:00

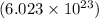 of particles.
of particles. contains
contains  contains
contains 


