In the first law of thermodynamics, △e = q-w, what does △e stand for?
a. the work done...

Chemistry, 09.01.2020 08:31 keigleyhannah30
In the first law of thermodynamics, △e = q-w, what does △e stand for?
a. the work done by the piston
b. the internal energy of the system
c. the heat added to the system
d. the change in the internal energy of the system

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2


Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2
You know the right answer?
Questions




English, 30.06.2019 10:30

English, 30.06.2019 10:30


Mathematics, 30.06.2019 10:30


Chemistry, 30.06.2019 10:30

Mathematics, 30.06.2019 10:30

Mathematics, 30.06.2019 10:30



Arts, 30.06.2019 10:30

Mathematics, 30.06.2019 10:30

Business, 30.06.2019 10:30



Social Studies, 30.06.2019 10:30

Social Studies, 30.06.2019 10:30

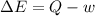
 represents the internal energy of the system.
represents the internal energy of the system.

