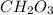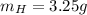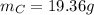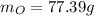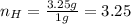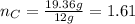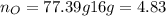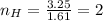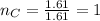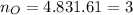Chemistry, 11.11.2019 22:31 mathisawsome123
What is the empirical formula of a compound composed of 3.25% hydrogen (h), 19.36% carbon (c), and 77.39% oxygen (o) by mass?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 22:30
Essay-alternative energy sources research sources of energy that are being developed. write a report of 350-400 words discussing the information you learned concerning the development of various energy sources and the impact that you think they will have on your life. include sources cited at the end of your report using the mla format. follow the rubric guidelines. note that wikipedia is not an appropriate resource for a research paper. worth 99
Answers: 3

Chemistry, 23.06.2019 02:00
What can be done to make a solid solute dissolve faster in a liquid solvent?
Answers: 1

Chemistry, 23.06.2019 04:10
In an experiment, 45g of silicon tetrachloride are treated with 45ml of water. what is the theoretical yield in grams of hcl
Answers: 3
You know the right answer?
What is the empirical formula of a compound composed of 3.25% hydrogen (h), 19.36% carbon (c), and 7...
Questions

Biology, 16.07.2019 13:20

History, 16.07.2019 13:20

History, 16.07.2019 13:20


English, 16.07.2019 13:20

Business, 16.07.2019 13:20

Mathematics, 16.07.2019 13:20


Social Studies, 16.07.2019 13:20



Biology, 16.07.2019 13:20


Mathematics, 16.07.2019 13:20

Mathematics, 16.07.2019 13:20


Chemistry, 16.07.2019 13:20

Chemistry, 16.07.2019 13:20

Mathematics, 16.07.2019 13:20

Mathematics, 16.07.2019 13:20