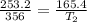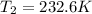Chemistry, 29.01.2020 08:51 hailiemanuel9362
Aballon has a volume of 253.2 ml. the volume of the balloon is decreased to 165.4 ml determine new tempature

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which part of a feedback mechanism is able to monitor the conditions outside of cells and usually uses nerve cells to relay this information to an intergrating center
Answers: 2

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1
You know the right answer?
Aballon has a volume of 253.2 ml. the volume of the balloon is decreased to 165.4 ml determine new t...
Questions




Social Studies, 21.08.2019 22:30

Mathematics, 21.08.2019 22:30

Chemistry, 21.08.2019 22:30

Mathematics, 21.08.2019 22:30

Geography, 21.08.2019 22:30

Mathematics, 21.08.2019 22:30

English, 21.08.2019 22:30



Mathematics, 21.08.2019 22:30


History, 21.08.2019 22:30

Mathematics, 21.08.2019 22:30


History, 21.08.2019 22:30


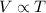 (At constant pressure and number of moles)
(At constant pressure and number of moles)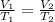
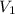 = initial volume of gas = 253.2 ml
= initial volume of gas = 253.2 ml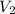 = final volume of gas = 165.4 ml
= final volume of gas = 165.4 ml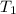 = initial temperature of gas= 356 K
= initial temperature of gas= 356 K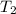 = final temperature of gas = ?
= final temperature of gas = ?