Chemistry, 28.12.2019 21:31 TrystinMarie2344
Which law is described by saying that when the pressure of a gas in a sealed container is cut in half, the gas will double in volume at a steady temperature?
boyle’s law
charles’s law
dalton’s law
gay-lussac’s law

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1

Chemistry, 23.06.2019 03:30
Which of the following describes the entropy change as a solution is made from a liquid and solid
Answers: 1
You know the right answer?
Which law is described by saying that when the pressure of a gas in a sealed container is cut in hal...
Questions

Computers and Technology, 18.08.2021 14:00

History, 18.08.2021 14:00

Mathematics, 18.08.2021 14:00




Computers and Technology, 18.08.2021 14:00

Mathematics, 18.08.2021 14:00






Mathematics, 18.08.2021 14:00

Mathematics, 18.08.2021 14:00


Biology, 18.08.2021 14:00

Mathematics, 18.08.2021 14:00

Mathematics, 18.08.2021 14:00

Social Studies, 18.08.2021 14:00

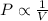
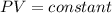 or
or