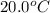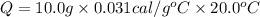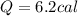Chemistry, 27.01.2020 02:31 chickennuggets0621
The specific heat of gold is 0.031 calories/gram°c. if 10.0 grams of gold were heated and the temperature of the sample changed by 20.0°c, how many calories of heat energy were absorbed by the sample?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2


Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3
You know the right answer?
The specific heat of gold is 0.031 calories/gram°c. if 10.0 grams of gold were heated and the temper...
Questions

Mathematics, 16.04.2021 02:10


Mathematics, 16.04.2021 02:10

Mathematics, 16.04.2021 02:10

Mathematics, 16.04.2021 02:10


Mathematics, 16.04.2021 02:10


Mathematics, 16.04.2021 02:10



Mathematics, 16.04.2021 02:10

Physics, 16.04.2021 02:10

Mathematics, 16.04.2021 02:10



Mathematics, 16.04.2021 02:20

Mathematics, 16.04.2021 02:20

Arts, 16.04.2021 02:20

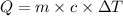
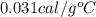
 = change in temperature =
= change in temperature =