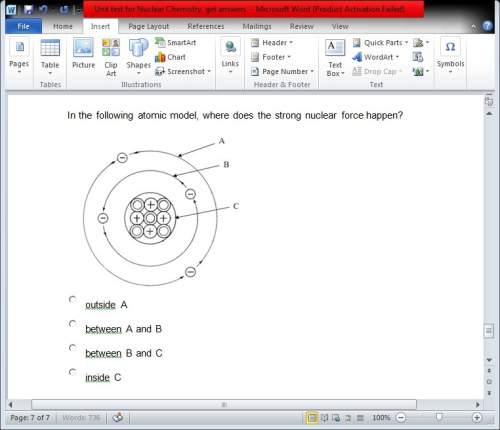
in the following atomic model, where does the strong nuclear force happen?
...


Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 23.06.2019 05:30
What is the body’s main processing system? it uses input from various parts to control voluntary and involutiontary movement. it’s composed of two main parts-the brain and spinal cord. a. nbs b.cns c. ans d. pns
Answers: 1

Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2

You know the right answer?
Questions

Health, 15.07.2019 14:30

Health, 15.07.2019 14:30



Mathematics, 15.07.2019 14:30



Mathematics, 15.07.2019 14:30

Health, 15.07.2019 14:30

Health, 15.07.2019 14:30

Mathematics, 15.07.2019 14:30




Computers and Technology, 15.07.2019 14:30

Computers and Technology, 15.07.2019 14:30


Computers and Technology, 15.07.2019 14:30

History, 15.07.2019 14:30

Computers and Technology, 15.07.2019 14:30