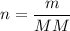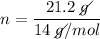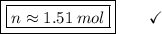Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1


You know the right answer?
If a sample contains 21.2 g n, how many moles of n does it contain? 0.66 mol 1.51 mol 14.01 mol 297...
Questions



Computers and Technology, 25.09.2021 20:40


English, 25.09.2021 20:40

Mathematics, 25.09.2021 20:40

Law, 25.09.2021 20:40


History, 25.09.2021 20:40





Biology, 25.09.2021 20:40

Mathematics, 25.09.2021 20:40



English, 25.09.2021 20:40

History, 25.09.2021 20:40