
Chemistry, 02.10.2019 18:30 maxraph108
Asolution is prepared by dissolving 23.7 g of cacl2 in 375 g of water. the density of the resulting solution is 1.05 g/ml. the concentration of cacl2 in this solution is molar.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:20
An aqueous solution of calcium hydroxide is standardized by titration with a 0.120 m solution of hydrobromic acid. if 16.5 ml of base are required to neutralize 27.5 ml of the acid, what is the molarity of the calcium hydroxide solution?
Answers: 3

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 06:30
Which of these natural resources is non-renewable a.corn b.wind c.geothermal d.natural gas
Answers: 2

You know the right answer?
Asolution is prepared by dissolving 23.7 g of cacl2 in 375 g of water. the density of the resulting...
Questions





Social Studies, 11.07.2019 21:00



Social Studies, 11.07.2019 21:00


Biology, 11.07.2019 21:00

Chemistry, 11.07.2019 21:00

Chemistry, 11.07.2019 21:00





Mathematics, 11.07.2019 21:00



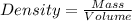
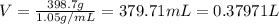
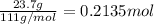
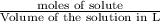
![[CaCl_2]=\frac{0.2135 mol}{0.37971 L}=0.5623 molar](/tpl/images/0283/7402/17e4b.png)


