
Chemistry, 18.11.2019 07:31 esdoles3865
If 5.65 grams of zinc metal react with 21.6 grams of silver nitrate, how many grams of silver metal can be formed and how many grams of the excess reactant will be left over when the reaction is complete? show all of your work.
unbalanced equation: zn + agno3 zn(no3)2 + ag

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Abullet found at a crime scene may be used as evidence in a trial if the percentage of metals match to the composition of metals in a bullet from the suspect's ammunition. a forensic scientist's analysis of the bullet shows that it contains 11.9 g of lead, 0.5 g of tin, and 0.8 b of antimony. what is the percentage of lead metal in the bullet? express your answers to the one's place.
Answers: 2


Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3
You know the right answer?
If 5.65 grams of zinc metal react with 21.6 grams of silver nitrate, how many grams of silver metal...
Questions




English, 17.10.2021 17:30


Advanced Placement (AP), 17.10.2021 17:30

Biology, 17.10.2021 17:30

English, 17.10.2021 17:30


Mathematics, 17.10.2021 17:30




Business, 17.10.2021 17:30

Mathematics, 17.10.2021 17:30

History, 17.10.2021 17:30

Social Studies, 17.10.2021 17:30



Mathematics, 17.10.2021 17:30

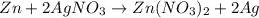
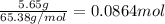
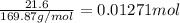
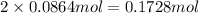 silver nitrate
silver nitrate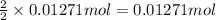 of silver metal
of silver metal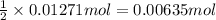 of zinc
of zinc

