Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 23:30
How many grams of ammonia would be produced by the decomposition of 16.93 mlof hydrazine? (the density of hydrazine is 1.021g/ml)
Answers: 3

Chemistry, 23.06.2019 04:00
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2

Chemistry, 23.06.2019 08:00
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
You know the right answer?
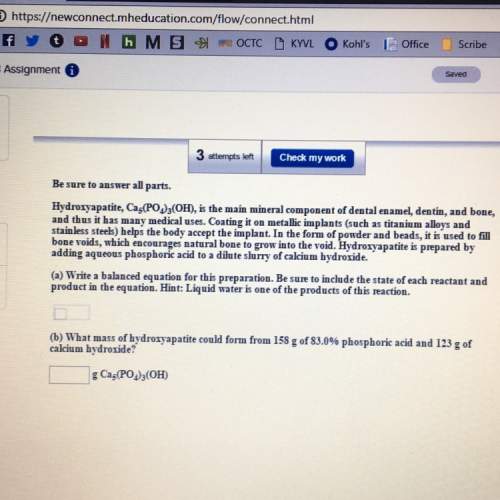
A) write a balanced equation. be sure to include the state of the reactant and product in the equati...
Questions


Mathematics, 12.01.2021 05:30

Social Studies, 12.01.2021 05:30


Advanced Placement (AP), 12.01.2021 05:30

Mathematics, 12.01.2021 05:30


Biology, 12.01.2021 05:30

Mathematics, 12.01.2021 05:30


English, 12.01.2021 05:40

Biology, 12.01.2021 05:40





Chemistry, 12.01.2021 05:40

Mathematics, 12.01.2021 05:40


English, 12.01.2021 05:40