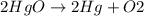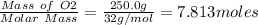Chemistry, 24.09.2019 11:30 vickygloom
Mercury(ii) oxide (hgo) decomposes to form mercury (hg) and oxygen (o2). the balanced chemical equation is shown below. 2hgo mc020-1.jpg 2hg + o2 the molar mass of hgo is 216.59 g/mol. the molar mass of o2 is 32.00 g/mol. how many moles of hgo are needed to produce 250.0 g of o2?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1


Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 23.06.2019 09:30
Organisms that live in the alpine and taiga biomes have developed unique adaptations that aid in their survival. moss campion is one of the plants found in the alpine biome. it has small leaves and a cushion shape that protect it from the wind and freezing temperatures in the alpine. how has the moss campion adapted to enable its survival in the alpine biome? a. waxy needles b. cone-shaped c. thin trunks d. low-growing
Answers: 1
You know the right answer?
Mercury(ii) oxide (hgo) decomposes to form mercury (hg) and oxygen (o2). the balanced chemical equat...
Questions




Chemistry, 28.09.2019 11:10



History, 28.09.2019 11:10

Mathematics, 28.09.2019 11:10

Biology, 28.09.2019 11:10

Mathematics, 28.09.2019 11:10

Chemistry, 28.09.2019 11:10


History, 28.09.2019 11:10


World Languages, 28.09.2019 11:10

History, 28.09.2019 11:10


Mathematics, 28.09.2019 11:10

World Languages, 28.09.2019 11:10

History, 28.09.2019 11:10