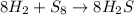Consider the balanced equation below. 8h2 + s8 mc006-1.jpg 8h2s based on the mole ratios, what can most likely be predicted? 1 mol of hydrogen will react with 1 mol of sulfur. 8 mol of hydrogen will react with 1 mol of sulfur. 8 mol of hydrogen will react with 8 mol of sulfur. 16 mol of hydrogen will react with 8 mol of sulfur.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 3

Chemistry, 23.06.2019 06:10
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1
You know the right answer?
Consider the balanced equation below. 8h2 + s8 mc006-1.jpg 8h2s based on the mole ratios, what can m...
Questions

Social Studies, 02.08.2019 08:00

Mathematics, 02.08.2019 08:00

World Languages, 02.08.2019 08:00

Biology, 02.08.2019 08:00

Spanish, 02.08.2019 08:00


Spanish, 02.08.2019 08:00

Mathematics, 02.08.2019 08:00










Social Studies, 02.08.2019 08:00

Geography, 02.08.2019 08:00

Social Studies, 02.08.2019 08:00