Chemistry, 01.10.2019 17:00 jet0120996
Magnesium burns in air with a dazzling brilliance to produce magnesium oxide: 2mg(s) + o2(g) ? 2mgo(s) when 4.00 g of magnesium burns, the theoretical yield of magnesium oxide is

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 22:20
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2

You know the right answer?
Magnesium burns in air with a dazzling brilliance to produce magnesium oxide: 2mg(s) + o2(g) ? 2mg...
Questions

Mathematics, 20.10.2019 18:20




Health, 20.10.2019 18:20

Mathematics, 20.10.2019 18:20

Chemistry, 20.10.2019 18:20




History, 20.10.2019 18:20


Biology, 20.10.2019 18:20


Health, 20.10.2019 18:20

Biology, 20.10.2019 18:20

Spanish, 20.10.2019 18:20


Physics, 20.10.2019 18:20

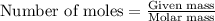 .....(1)
.....(1)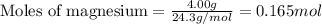
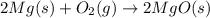
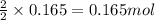 of magnesium oxide
of magnesium oxide