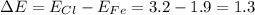Chemistry, 20.12.2019 22:31 mbrisen3396
The bond formed between iron (electronegativity = 1.9) and chlorine (electronegativity = 3.2) is

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1

Chemistry, 23.06.2019 06:00
In an exothermic reaction at equilibrium, what is the effect of lowering the temperature? a. the reaction makes more products. b. the reaction makes more reactants. c. the reaction is unchanged.
Answers: 1

Chemistry, 23.06.2019 10:30
Silver is a white metal that is an excellent conductor. silver tarnishes when exposed to air and light. the density of silver is 10.49 g/cm3. the melting point is 962oc and the boiling point is 2000oc. a chemical property of silver is
Answers: 3
You know the right answer?
The bond formed between iron (electronegativity = 1.9) and chlorine (electronegativity = 3.2) is...
Questions

Arts, 04.02.2021 17:50


Mathematics, 04.02.2021 17:50

Social Studies, 04.02.2021 17:50

Biology, 04.02.2021 17:50

Mathematics, 04.02.2021 17:50

Biology, 04.02.2021 17:50

Mathematics, 04.02.2021 17:50


Engineering, 04.02.2021 17:50



Mathematics, 04.02.2021 17:50

Mathematics, 04.02.2021 17:50

Mathematics, 04.02.2021 17:50

Mathematics, 04.02.2021 17:50

Mathematics, 04.02.2021 17:50

English, 04.02.2021 17:50

Mathematics, 04.02.2021 17:50

English, 04.02.2021 17:50