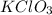Chemistry, 23.09.2019 11:30 nauticatyson9
A13.00 g sample of a compound contains 4.15 g potassium (k), 3.76 g chlorine (cl), and oxygen (o). calculate the empirical formula.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Follow the steps provided in the simulation to add water to the graduated cylinder, select one of the three samples (copper, silver, or gold), set its mass to the values given in the statements below, and calculate its density. here is a summary of the steps required: add water by clicking and holding prepare a known volume of water button. until the desired volume of water has been added. if more than the desired volume is added, click the reset button. button and redo the procedure. a single click will add about 21.0 ml of water. to set the mass, click and hold weigh out metal button. until the desired amount of metal is added to the weighing pan. once the desired mass of the metal is added, release the button. transfer the metal to water and then click on calculate density button. to see how the density is calculated using water displacement to measure the volume of the solid. to save time you can approximate the initial volume of water to â±1 ml and the initial mass of the solid to â±1 g. for example, if you are asked to add 23 ml of water, add between 22 ml and 24 ml. which metals in each of the following sets will have equal density? check all that apply.
Answers: 1


Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1
You know the right answer?
A13.00 g sample of a compound contains 4.15 g potassium (k), 3.76 g chlorine (cl), and oxygen (o). c...
Questions



Biology, 01.11.2020 23:50

Mathematics, 01.11.2020 23:50

English, 01.11.2020 23:50

Mathematics, 01.11.2020 23:50

English, 01.11.2020 23:50


History, 01.11.2020 23:50



Chemistry, 01.11.2020 23:50


Mathematics, 01.11.2020 23:50


Arts, 01.11.2020 23:50

Biology, 01.11.2020 23:50

Arts, 01.11.2020 23:50

Mathematics, 01.11.2020 23:50

English, 01.11.2020 23:50