
Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:10
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 03:30
The molar mass of nickel(ni) is 58.7 g/mol. how many moles are in an 88 gram sample of nickel?
Answers: 1
You know the right answer?
Calculate the mass (in ng) of 2.33 x 1020 atoms of oxygen....
Questions

Physics, 19.07.2019 22:00



Mathematics, 19.07.2019 22:00

Mathematics, 19.07.2019 22:00

Biology, 19.07.2019 22:00

Mathematics, 19.07.2019 22:00

Mathematics, 19.07.2019 22:00

History, 19.07.2019 22:00

Mathematics, 19.07.2019 22:00


Physics, 19.07.2019 22:00

English, 19.07.2019 22:00




Mathematics, 19.07.2019 22:00


Mathematics, 19.07.2019 22:00

Social Studies, 19.07.2019 22:00

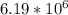 g of Oxygen
g of Oxygen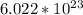
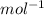
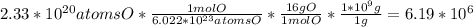 g of Oxygen
g of Oxygen

