
Chemistry, 23.09.2019 21:00 alyxkellar06
For the reaction c2h6 (g) → c2h4 (g) + h2 (g) δh° is +137 kj/mol and δs° is +120 j/k ∙ mol. this reaction is question 10 options: spontaneous only at high temperature spontaneous at all temperatures spontaneous only at low temperature nonspontaneous at all temperatures

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1

Chemistry, 23.06.2019 00:00
If many scientists conduct the same or similar experiments, and all obtain similar results, a can be written, which is a generally agreed-upon statement that explains and predicts how a natural phenomenon works.
Answers: 1

Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1
You know the right answer?
For the reaction c2h6 (g) → c2h4 (g) + h2 (g) δh° is +137 kj/mol and δs° is +120 j/k ∙ mol. this rea...
Questions


History, 04.11.2020 20:30


Mathematics, 04.11.2020 20:30

Health, 04.11.2020 20:30


Mathematics, 04.11.2020 20:30

Mathematics, 04.11.2020 20:30

Social Studies, 04.11.2020 20:30


History, 04.11.2020 20:30


Mathematics, 04.11.2020 20:30

Mathematics, 04.11.2020 20:30

English, 04.11.2020 20:30


Mathematics, 04.11.2020 20:30

Chemistry, 04.11.2020 20:30


Mathematics, 04.11.2020 20:30

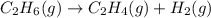
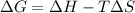
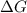 = Gibbs free energy
= Gibbs free energy  = +ve, reaction is non spontaneous
= +ve, reaction is non spontaneous
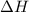 = enthalpy change = endothermic = +137 KJ
= enthalpy change = endothermic = +137 KJ 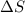 = entropy change = +120 J/K
= entropy change = +120 J/K 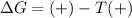
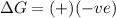
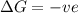 when
when  has more value than
has more value than 

