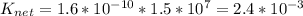The solubility of silver chloride can be increased by dissolving it in a solution containing ammonia. agcl (s) ag+ (aq) + cl- (aq) k1 = 1.6 x 10-10 ag+ (aq) + 2nh3 (aq) ag(nh3)2+ (aq) k2 = 1.5 x 107 what is the value of the equilibrium constant for the overall reaction? agcl (s) + 2nh3 (aq) ag(nh3)2+ (aq) + cl- (aq) knet = ?

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1

Chemistry, 23.06.2019 07:50
Asolution is produced in which water is the solvent and there are four solutes. which of the solutes can dissolve better if the solution is heated?
Answers: 1

Chemistry, 23.06.2019 08:00
Amechanical wave that transports a lot of energy will have a
Answers: 2
You know the right answer?
The solubility of silver chloride can be increased by dissolving it in a solution containing ammonia...
Questions

Biology, 04.02.2020 03:57

Arts, 04.02.2020 03:57

Mathematics, 04.02.2020 03:57

English, 04.02.2020 03:57

Mathematics, 04.02.2020 03:57

Mathematics, 04.02.2020 03:57



Business, 04.02.2020 03:57

Mathematics, 04.02.2020 03:57

Mathematics, 04.02.2020 03:57

History, 04.02.2020 03:57





Mathematics, 04.02.2020 03:57

English, 04.02.2020 03:57

Social Studies, 04.02.2020 03:57

![K_{net}=[Cl^-]*[Ag(NH_3)_2^{+2}]=2.4*10^{-3}](/tpl/images/0309/4158/aeebe.png)
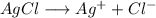
![K_1=[Ag^+]*[Cl^-]](/tpl/images/0309/4158/2f3df.png)

![K_2=\frac{[Ag(NH_3)_2^{+2}]}{[Ag^+]}](/tpl/images/0309/4158/d8bb8.png)
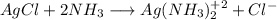
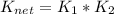
![K_{net}=[Ag^+]*[Cl^-]*\frac{[Ag(NH_3)_2^{+2}]}{[Ag^+]}](/tpl/images/0309/4158/ccfbe.png)
![K_{net}=[Cl^-]*[Ag(NH_3)_2^{+2}]](/tpl/images/0309/4158/2d974.png)