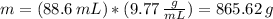Chemistry, 30.08.2019 20:00 hallmansean04
Determine the mass of an object that has a volume of 88.6 ml and a density of 9.77 g/ml.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3


Chemistry, 23.06.2019 06:00
Nthis lab, you will do experiments to identify types of changes. using the question format you learned (shown above), write an investigative question that you can answer by doing these experiments
Answers: 3
You know the right answer?
Determine the mass of an object that has a volume of 88.6 ml and a density of 9.77 g/ml....
Questions

Geography, 13.04.2020 14:52



Social Studies, 13.04.2020 14:52


History, 13.04.2020 14:52


Mathematics, 13.04.2020 14:52


Mathematics, 13.04.2020 14:54


Mathematics, 13.04.2020 14:54



Health, 13.04.2020 15:13


Mathematics, 13.04.2020 15:14

English, 13.04.2020 15:14


Social Studies, 13.04.2020 15:15