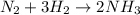Chemistry, 31.08.2019 16:30 icashaypabozp
In 1912, chemist fritz haber developed a process that combined nitrogen from the air with hydrogen at high temperatures and pressures to make ammonia. specifically, the process involved combining one molecule of nitrogen gas (n2) with three molecules of hydrogen gas (h2) to get two molecules of ammonia (nh3). if you write this process in a symbol format, it looks like this:
n2 + 3h2 → 2nh3
explain whether this is a chemical or physical change, and why. does it involve elements, compounds, mixtures, or pure substances? also describe how many atoms are involved before and after. what do you notice about the number of atoms?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 23.06.2019 06:30
Which of the following steps is not likely to take place during cellular respiration? (5 points) select one: a. oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. c. simple sugar breaks down. d. energy is used up.
Answers: 1

Chemistry, 23.06.2019 11:20
When using the ideal gas law constant 0.0821, what unit is used for volume? a) galloonb) ouncec) milliliterd) liter
Answers: 1
You know the right answer?
In 1912, chemist fritz haber developed a process that combined nitrogen from the air with hydrogen a...
Questions



Mathematics, 13.10.2020 06:01

English, 13.10.2020 06:01


Mathematics, 13.10.2020 06:01

Computers and Technology, 13.10.2020 06:01

History, 13.10.2020 06:01

Spanish, 13.10.2020 06:01

History, 13.10.2020 06:01

Mathematics, 13.10.2020 06:01

Mathematics, 13.10.2020 06:01



Chemistry, 13.10.2020 06:01

Mathematics, 13.10.2020 06:01

Computers and Technology, 13.10.2020 06:01

Mathematics, 13.10.2020 06:01