Which statement best describes a molecular formula?
a. a molecular formula gives only...

Chemistry, 15.10.2019 20:40 tiffanibell71
Which statement best describes a molecular formula?
a. a molecular formula gives only the position of atoms present in a molecule
b. a molecular formula gives only the number and kind of atoms present in a molecule
c. a molecular formula gives only the number and kind of atoms present in a molecule and shows how large each atom is.
d. a molecular formula gives only the size of the atoms present in a molecule.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1
You know the right answer?
Questions







Computers and Technology, 26.09.2019 20:00













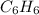 which gives the actual number of atoms of each element present and the empirical formula of benzene is
which gives the actual number of atoms of each element present and the empirical formula of benzene is 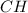 which gives the simplest ratio of atoms present in it.
which gives the simplest ratio of atoms present in it.

