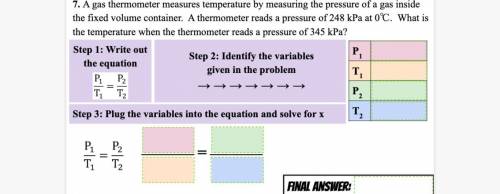
This question down there!
...

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1
You know the right answer?
Questions

Mathematics, 28.01.2020 15:54

Social Studies, 28.01.2020 15:54

English, 28.01.2020 15:54

Mathematics, 28.01.2020 15:54



Geography, 28.01.2020 15:54

History, 28.01.2020 15:54

Biology, 28.01.2020 15:54



History, 28.01.2020 15:54


Physics, 28.01.2020 15:54


Computers and Technology, 28.01.2020 15:54

English, 28.01.2020 15:54

Physics, 28.01.2020 15:54


English, 28.01.2020 15:54