
Chemistry, 27.04.2021 20:50 freshysans4
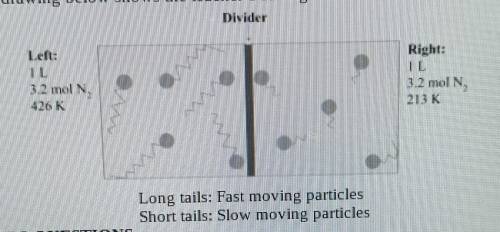
A teacher sets up a box containing 2 identical halves by inserting a divider in the middle of the box. Both the box and the divider are made of an insulating material that does not absorb or give 3.2 moles of N² gas. The temperature of the gas in the left compartment is 426 K, and in the right one 213 K. The drawing below shows the teacher's setting.
Answer The 3 Questions:
1.What is the gas pressure in the left chamber
2.The pressure in the right chamber is half the pressure of the left chamber. Explain why this is so using the Kinetic Theory of Glases.
3.What do you estimate will be the final gas temperature after removing the divider? Explain your results using the Kinetic Theory of Gases.

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 07:40
)in the deacon process for the manufacture of chlorine, hcl and o2 react to form cl2 and h2o. sufficient air (21 mole% o2, 79% n2) is fed to provide 35% excess oxygen, and the fractional conversion of hcl is 85%. calculate the mole fractions of the product stream components.
Answers: 1

Chemistry, 23.06.2019 09:00
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate according to the following equation:
Answers: 2

Chemistry, 23.06.2019 13:00
Me puede ayudar con estas preguntas 1. diga que estudia la química orgánica explica el nacimiento bioquímicas 2. cual es la importancia de la química orgánica. 3. determine las principales características del hidrógeno, oxigeno, nitrógeno y azufre como elementos que constituyen los compuestos orgánicas. 4. elabore una tabla comparativa entre compuestas orgánicas e incaicos.
Answers: 1
You know the right answer?
A teacher sets up a box containing 2 identical halves by inserting a divider in the middle of the bo...
Questions

Social Studies, 24.04.2020 18:53

Computers and Technology, 24.04.2020 18:53


Spanish, 24.04.2020 18:53




Biology, 24.04.2020 18:54





Mathematics, 24.04.2020 18:54


Chemistry, 24.04.2020 18:54

Biology, 24.04.2020 18:54


English, 24.04.2020 18:54

English, 24.04.2020 18:54



