
Chemistry, 29.04.2021 18:30 historyfanboy101
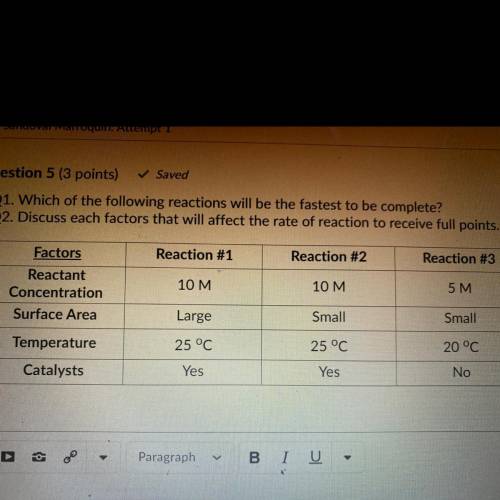
Q1. Which of the following reactions will be the fastest to be complete? Q2. Discuss each factors that will affect the rate of reaction to receive full points.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3
You know the right answer?
Q1. Which of the following reactions will be the fastest to be complete?
Q2. Discuss each factors...
Questions

Mathematics, 18.03.2021 23:00


Mathematics, 18.03.2021 23:00

Mathematics, 18.03.2021 23:00



History, 18.03.2021 23:00

Business, 18.03.2021 23:00

Mathematics, 18.03.2021 23:00





History, 18.03.2021 23:00


English, 18.03.2021 23:00






