Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1

Chemistry, 22.06.2019 23:30
If it is an isoelectronic series select true, if not select false. o2-, s2-, se2-, te2- na+, k+, rb+, cs+ n3-, p3-, as3-, sb3- ag, cd+, sn3+, sb4+ f-, cl-, br-, i- f-, ne, na+, mg2+ s2-, s, s6+
Answers: 1
You know the right answer?
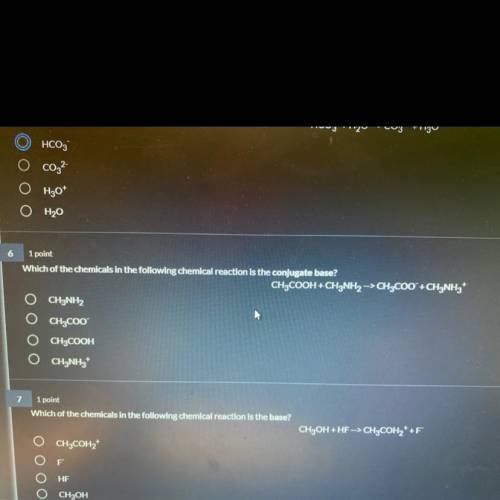
Which of the chemicals in the following chemical reaction is the conjugate base?
CH3COOH + CH3NH2...
Questions

Mathematics, 22.02.2021 08:50


History, 22.02.2021 08:50

Mathematics, 22.02.2021 08:50

Mathematics, 22.02.2021 08:50





Mathematics, 22.02.2021 08:50

World Languages, 22.02.2021 08:50


Chemistry, 22.02.2021 08:50

Mathematics, 22.02.2021 08:50

Social Studies, 22.02.2021 08:50

Mathematics, 22.02.2021 08:50

Mathematics, 22.02.2021 08:50

Physics, 22.02.2021 08:50