
Chemistry, 30.04.2021 15:40 fatboicroi
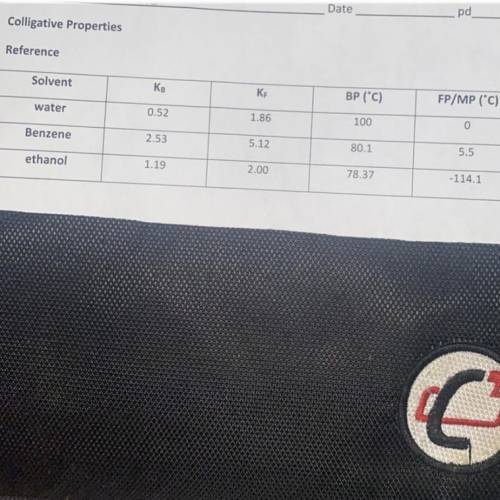
When 25.5 grams of a molecular substance is dissolved in 225g benzene, the solution begins to freeze at -5.05C. Calculate the molar mass of this solute (I need to understand the work, and look at the image attached for the key for benzene.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1

Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3
You know the right answer?
When 25.5 grams of a molecular substance is dissolved in 225g benzene, the solution begins to freeze...
Questions

Mathematics, 09.09.2019 18:20

Physics, 09.09.2019 18:20




Mathematics, 09.09.2019 18:20



Mathematics, 09.09.2019 18:20



Mathematics, 09.09.2019 18:20

Business, 09.09.2019 18:20


History, 09.09.2019 18:20




Mathematics, 09.09.2019 18:20

Mathematics, 09.09.2019 18:20



