Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 22.06.2019 21:30
What is the correct name for the compound cocl3? a) cobalt(i) chloride b) cobalt(i) chlorate c) cobalt(ii) chlorate d) cobalt(iii) chloride
Answers: 1
You know the right answer?
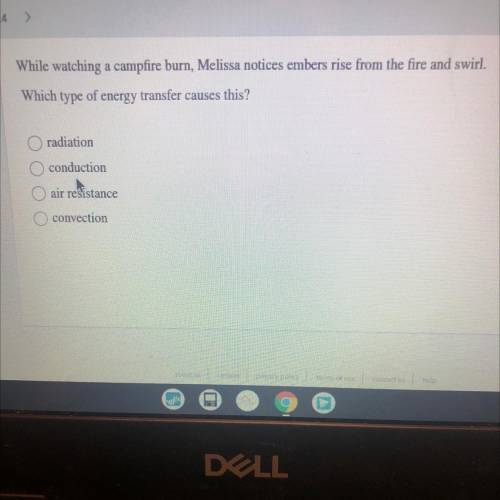
While watching a campfire burn, Melissa notices embers rise from the fire and swirl.
Which type of...
Questions


English, 06.12.2019 02:31

Biology, 06.12.2019 02:31



History, 06.12.2019 02:31

English, 06.12.2019 02:31

Biology, 06.12.2019 02:31

History, 06.12.2019 02:31


Mathematics, 06.12.2019 02:31

Mathematics, 06.12.2019 02:31

Mathematics, 06.12.2019 02:31

Mathematics, 06.12.2019 02:31



Mathematics, 06.12.2019 02:31


English, 06.12.2019 02:31

History, 06.12.2019 02:31