
Chemistry, 05.05.2021 21:40 jasperzhouzihe3018
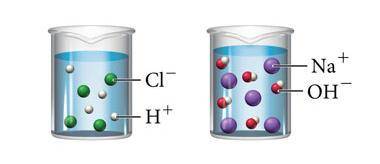
The image above shows what happens when hydrochloric acid (HCl) and sodium hydroxide (NaOH) are mixed with water. Which statement below is true about both solutions?
A. Both solutions contain solutes that do not dissolve completely in water.
B. Both solutions are saturated because they contain the maximum concentration of a solute dissolved in the solvent.
C. Both solutions will conduct an electric current because they both contain ions that can carry a charge.
D. Both solutions are acidic because they both contain hydrogen.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 23.06.2019 03:00
The size (radius) of an oxygen molecule is about 2.0 ×10−10m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
Answers: 3
You know the right answer?
The image above shows what happens when hydrochloric acid (HCl) and sodium hydroxide (NaOH) are mixe...
Questions

English, 21.10.2020 04:01

SAT, 21.10.2020 04:01

Mathematics, 21.10.2020 04:01


Mathematics, 21.10.2020 04:01


English, 21.10.2020 04:01

History, 21.10.2020 04:01

Advanced Placement (AP), 21.10.2020 04:01

Mathematics, 21.10.2020 04:01

Mathematics, 21.10.2020 04:01

Mathematics, 21.10.2020 04:01

Advanced Placement (AP), 21.10.2020 04:01

Mathematics, 21.10.2020 04:01


English, 21.10.2020 04:01

Mathematics, 21.10.2020 04:01


Chemistry, 21.10.2020 04:01




