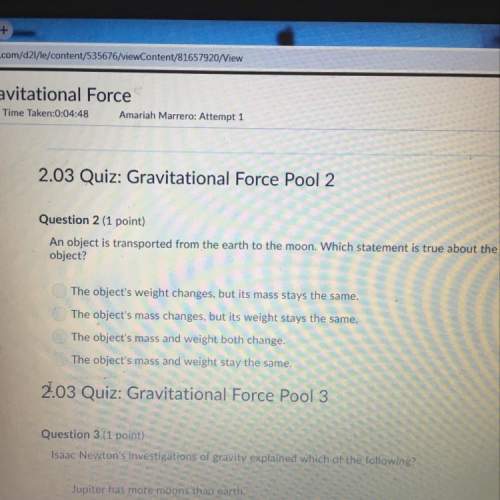
Chemistry, 06.05.2021 06:40 mattie2353
Using the standard enthalpies of formation provided,
calculate the standard enthalpy for the following reaction.
NaOH(s) + HCl(g) > NaCl(s) + H2O(g)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Agroup of students is studying convection currents. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other in an area with warm air. after 10 minutes, the balloons are released from a height of 1 meter. which of the following do the students most likely observe? a. the balloons both rise. the cold balloon is larger than the warm balloon. b. the balloons rise at the same rate. both balloons are the same size. c. the warm balloon expands and rises. the cold balloon shrinks and sinks. d. the cold balloon expands and rises. the warm balloon shrinks and sinks.
Answers: 2

Chemistry, 22.06.2019 05:20
Asolution contains 180 g of glucose (c6h12o6) and 162 g of water. what is the mole fraction of glucose?
Answers: 3

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3
You know the right answer?
Using the standard enthalpies of formation provided,
calculate the standard enthalpy for the follo...
Questions




History, 20.01.2020 08:31


History, 20.01.2020 08:31




Chemistry, 20.01.2020 08:31

Mathematics, 20.01.2020 08:31

English, 20.01.2020 08:31

Geography, 20.01.2020 08:31

Mathematics, 20.01.2020 08:31

Health, 20.01.2020 08:31

Mathematics, 20.01.2020 08:31

Health, 20.01.2020 08:31


Mathematics, 20.01.2020 08:31