
Chemistry, 07.05.2021 14:00 tmcdowell69
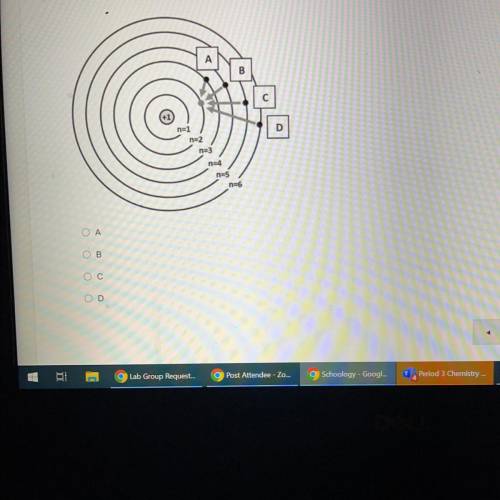
Which of the electron movements shown below (A, B, C, or D) is most likely to produce a red-colored light?
B
D
na3
n24
n25
n=6
Ο Α
OB
Ос
OD

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
Which of the electron movements shown below (A, B, C, or D) is most likely to produce a red-colored...
Questions




Computers and Technology, 15.05.2021 23:40


Mathematics, 15.05.2021 23:40





Chemistry, 15.05.2021 23:40


Mathematics, 15.05.2021 23:40

Mathematics, 15.05.2021 23:40

English, 15.05.2021 23:40




Mathematics, 15.05.2021 23:40




