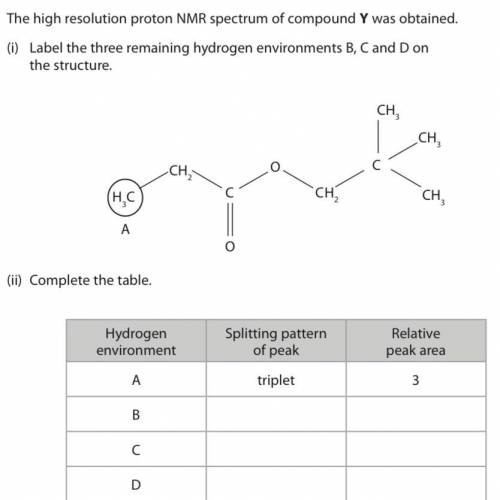
Can someone please help me please?
Thanks in advance
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 17:10
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1

Chemistry, 23.06.2019 09:00
A0.10 m aqueous solution of sodium sulfate is a better conductor of electricity than a 0.10 m aqueous solution of sodium chloride. which of the following best explains this observation? (a) sodium sulfate is more soluble in water than sodium chloride. (b) sodium sulfate has a higher molar mass than sodium chloride. (c) to prepare a given volume of 0.10 m solution, the mass of sodium sulfate needed is more than twice the mass of sodium chloride needed. (d) more moles of ions are present in a given volume of 0.10 m sodium sulfate than in the same volume of 0.10 m sodium chloride. (e) the degree of dissociation of sodium sulfate in solution is significantly greater than that of sodium chloride.
Answers: 2

Chemistry, 23.06.2019 11:30
If this sedimentary rock layer is truly the oldest one of marine origin, what do you think that tells usabout the formation of earth's oceans?
Answers: 2
You know the right answer?
Questions

Chemistry, 04.08.2019 06:30

History, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30


Physics, 04.08.2019 06:30

Spanish, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30

Spanish, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30


Spanish, 04.08.2019 06:30

Social Studies, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30


Health, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30

Social Studies, 04.08.2019 06:30

Social Studies, 04.08.2019 06:30

History, 04.08.2019 06:30