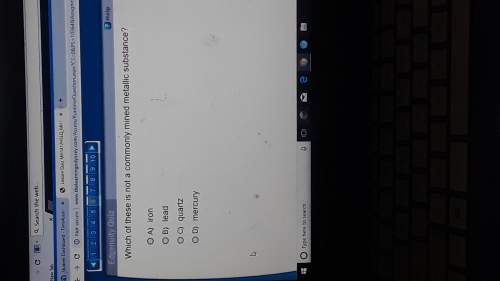
On last question, could use help
Which of the following is true for a gas under conditions of very high pressure?
PV > nRT, because the real volume of the gas would be more than the ideal volume.
PV = nRT, because intermolecular forces are considerable at very high pressures.
PV = nRT, because all gases behave as ideal gases at very high pressures.
PV < nRT, because the volume of the gas would become negligible.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:00
An example of technology is the a. addition of a side group to an organic molecule during synthesis. b. use of a new antibiotic to fight an infection. c. measurement of iron concentration in a water sample. d. study of atomic fusion reactions
Answers: 3

Chemistry, 21.06.2019 18:50
Problem page gaseous ethane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . if of water is produced from the reaction of of ethane and of oxygen gas, calculate the percent yield of water. be sure your answer has the correct number of significant digits in it.
Answers: 2

Chemistry, 21.06.2019 20:30
Which of the following pairs of elements belong to the same groupa. h and he b. li and bec. c and pb d. ga and ge
Answers: 1

Chemistry, 22.06.2019 01:00
Consider three unlabeled bottles, each contain small pieces of one of the following metals. - magnesium - sodium - silver the following reagents are used for identifying the metals. - pure water - a solution of 1.0 molar hcl - a solution of concentrated hno3 (a) which metal can be easily identified because it is much softer than the other two? describe a chemical test that distinguishes this metal from the other two, using only one of the reagents above. write a balanced chemical equation for the reaction that occurs. (b) one of the other two metals reacts readily with the hcl solution. identify the metal and write the balanced chemical equation for the reaction that occurs when this metal is added to the hcl solution. use the table of standard reduction potentials (attached) to account for the fact that this metal reacts with hcl while the other does not. (c) the one remaining metal reacts with the concentrated hno3 solution. write a balanced chemical equation for the reaction that occurs. (d) the solution obtained in (c) is diluted and a few drops of 1 m hcl is added. describe what would be observed. write a balanced chemical equation for the reaction that occurs.
Answers: 2
You know the right answer?
On last question, could use help
Which of the following is true for a gas under conditions of very...
Questions

English, 13.01.2020 09:31


Mathematics, 13.01.2020 09:31


History, 13.01.2020 09:31

Social Studies, 13.01.2020 09:31

Mathematics, 13.01.2020 09:31

Biology, 13.01.2020 09:31


English, 13.01.2020 09:31


Chemistry, 13.01.2020 09:31

Mathematics, 13.01.2020 09:31




Physics, 13.01.2020 09:31


Mathematics, 13.01.2020 09:31