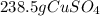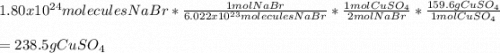Chemistry, 12.05.2021 02:10 BaileyElizabethRay
CuSO4 + 2NaBr --> CuBr2 + Na2SO4 ? How many grams of CuSO4 are needed to react with 1.80 x 1024 molecules of NaBr?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1
You know the right answer?
CuSO4 + 2NaBr --> CuBr2 + Na2SO4 ? How many grams of CuSO4 are needed to react with 1.80 x 1024 m...
Questions

English, 12.09.2019 18:30

History, 12.09.2019 18:30

Mathematics, 12.09.2019 18:30









Spanish, 12.09.2019 18:30



Spanish, 12.09.2019 18:30



Mathematics, 12.09.2019 18:30